Άκης
Administrator
Effects of Revivogen Scalp Therapy on Testosterone Metabolism in Reconstructed Human Epidermis
STUDY REPORT AD070315B-2
Study client: ADVANCED SKIN AND HAIR Inc.
Dr. Alex Khadavi, MD
12121 Wilshire Blvd
Suite 1012
LOS ANGELES CA 90025
USA
More info: go to Revivogen
Study n°AD070315B
Effects of Revivogen Scalp Therapy
ADVANCED SKIN AND HAIR Inc. on testosterone metabolism in reconstructed human epidermis
The investigators and the author of this report hereby certify the validity of the data presented and attest their full agreement with the conclusions presented at the end of the report.
Certified by:
Name: Franck JUCHAUX
Position: Study director
Date: October 18th, 2007 Signature
TABLE OF CONTENT
1. Introduction
2. Materials & Methods
2.1 Biological Model
2.2 Test Compound & References
2.3 Testosterone
2.4 Treatment
2.5 Extractions & Analysis
3. Results & Conclusion
3.1 RHE Viability
3.2 Testosterone Metabolism
4. Tables & Figures
1 - INTRODUCTION
ADVANCED SKIN AND HAIR, INC. has developed the compound Revivogen Scalp Therapy for hair loss. Male pattern hair loss (MPHL) is a potentially reversible condition in which dihydrotestosterone is an important etiologic factor (Olsen et al, 2006). Dihydrotestosterone, the efficient steroid, is produced from testosterone by 5α reductase. Inhibition of 5α reductase activity is known to improve MPHL.
Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, Wilson T, Rittmaster RS, 2006. The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. J Am Acad Dermatol., 55(6):1014-23
BIOalternatives performed this study in order to assess the effects of the test compound on the metabolism of testosterone in reconstructed human epidermis. This model has been shown to be useful for the evaluation of inhibitors of this metabolism (Bernard et al. 2000; Int. J. Cosm. Sci.,22, 397-407).
The steroid 5α-reductase isoenzymes (5αR) transform testosterone (T) into 17β-hydroxy-5α-androstan-3-one (5-dihydrotestosterone, DHT). This reaction is crucial in the action of androgens.

2 - MATERIALS AND METHODS 2.1 Biological model
Reconstructed Human Epidermis (RHE)
- Tissues: 18 RHE (0.50 cm², 10 days), batch n° 01015-31
- Culture: at 37°C and 5 % CO2
- Culture medium: differentiation medium
2.2 Test compound and references
Test compound Stock-solution Dilution Application
Revivogen Scalp Therapy
batch 07-06 (AD070315/2) Liquid supplied by the study promoter and stored at room temperature. - topical at 50 µl/RHE
References Stock-solution Dilution Application
Finasteride
batch 231664 10-2 M in ethanol In water topical at 10-5 M/RHE
Avodart®
batch 053721A 10-3 M in ethanol In water topical at 10-6 and 10-5 M/RHE
2.3 Testosterone
Testosterone: [4-14C] testosterone (Amersham B76, 54 mCi/mmole, 2.35 nmole/epidermis). [4-14C] testosterone stock-solution was dissolved in ethanol and diluted in sterile water (1% ethanol final).
2.4 Treatment
The RHE were topically treated (or not, control) with the test compound or the references. Three RHE were used for each experimental condition.
After 24h of treatment, the RHE were topically re-treated and incubated for 5 hours. After incubation, the test compound and references were removed from the top of the RHE and 100 µl of the labelled testosterone solution were loaded on the stratum corneum of each RHE (127 nCi/epidermis).
After a 24-hour incubation period, the media underneath the RHE were collected for sterols analysis.
2.5 Extractions and analysis
Transepidermal diffusion assessment: the amount of testosterone that passed through the epidermal tissues was measured by liquid scintillation counting (LKB 1211 Rackbeta counter) of a fraction of culture medium.
Metabolism analysis: the steroid molecules from culture media were extracted by 2 volumes of chloroform/methanol (98:2) and dried. The various molecular species (testosterone metabolites) were separated by thin layer chromatography (TLC) on silica plates (RE/Silice, Whatman) in a solvent system containing dichloromethane, ethylacetate and methanol (85:15:3). The plates were autoradiographed and testosterone metabolites were quantified using a phosphorImager and specific software (Packard instrument).
3 - RESULTS AND CONCLUSION
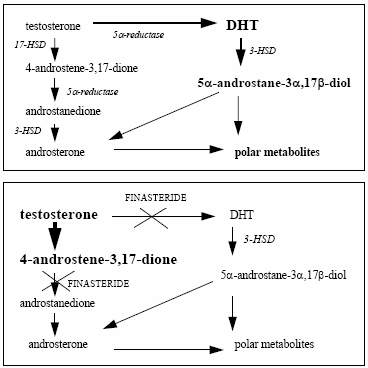
Schematic simplified pathway for testosterone metabolism.
Effects of finasteride (from Bernard F-X et al., Int. J. Cosmetic Science, 22 397-407 (2000))
Tables 1 and 2
Untreated control:
After 24h of culture, the rate of testosterone metabolism was very high.
Dihydrotestosterone (DHT) was clearly identified in the steroid profile. DHT was the major metabolite in the control epidermis. After 24h, about 74% of the deposited testosterone was converted into DHT. Other important metabolites were androstane-diols (e) and 4-androstene-3,17-dione (b).
Effects of finasteride:
Finasteride at 10-5 M strongly inhibited the transformation of testosterone into DHT (67% inhibition compared to the control). Furthermore, as expected, finasteride decreased the amount of androstane-diols (e) and induced a strong accumulation of 4-androstene-3,17-dione (b) (Figure 1).
Effects of dutasteride:
Dutasteride at 10-6 M and 10-5 M strongly inhibited dose dependently the transformation of testosterone into DHT (respectively 80% and 86% of inhibition of the DHT production compared to the control). Furthermore, as expected, dutasteride decreased the amount of androstane-diols (e) and induced a strong accumulation of 4-androstene-3,17-dione (b) (Figure 1).
Effects of Revivogen Scalp Therapy:
Revivogen Scalp Therapy (5 mg/cm²) strongly reduced the production of DHT (90% of
inhibition of the DHT production compared to the control).
4 - TABLES AND FIGURES
Table 1: Diffusion of [14C]-testosterone (and metabolites) through RHE. Trans-epidermal diffusion (24h)
Treatment Conc cpm sd n % control nmol steroid
Total testosterone - 326060 - 1 - 2,35
Unrelated control - 125307 14977 3 100 0,9
Finasteride 10(-5)M 147747 6335 3 118 1,1
Dutasteride 10(-5)M
10(-6)M 150160
159213 10809
20653 3
3 120
127 1,1
1,1
Revivogen Scalp Therapy 5 mg/cm2 30026 1181 3 24 0,2
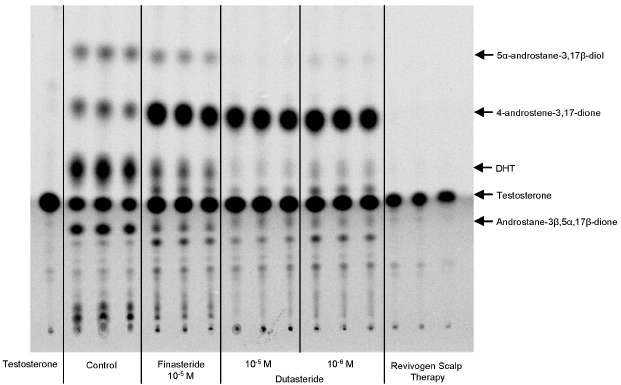
Figure 1: Thin layer chromatography and autoradiography of [14C]-testosterone and metabolites after transepidermal diffusion (24h).
Table 2: Effects of Revivogen Scalp Therapy and the reference compounds on the production of testosterone metabolites. Instant Imager analysis of TLC in figure 2 (direct radioactivity measurement)
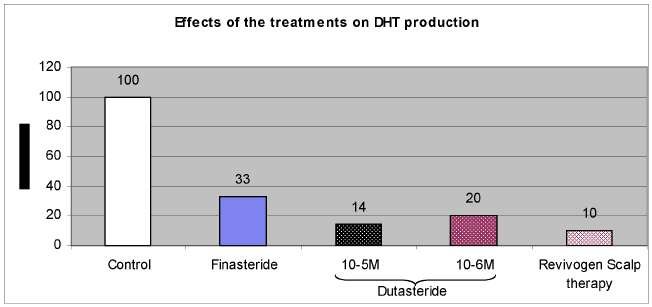
Figure 2: Effects of Revivogen Scalp Therapy and the reference compounds on DHT metabolism.
[H έρευνα είναι attached στο παρών post]
STUDY REPORT AD070315B-2
Study client: ADVANCED SKIN AND HAIR Inc.
Dr. Alex Khadavi, MD
12121 Wilshire Blvd
Suite 1012
LOS ANGELES CA 90025
USA
More info: go to Revivogen
Study n°AD070315B
Effects of Revivogen Scalp Therapy
ADVANCED SKIN AND HAIR Inc. on testosterone metabolism in reconstructed human epidermis
The investigators and the author of this report hereby certify the validity of the data presented and attest their full agreement with the conclusions presented at the end of the report.
Certified by:
Name: Franck JUCHAUX
Position: Study director
Date: October 18th, 2007 Signature
TABLE OF CONTENT
1. Introduction
2. Materials & Methods
2.1 Biological Model
2.2 Test Compound & References
2.3 Testosterone
2.4 Treatment
2.5 Extractions & Analysis
3. Results & Conclusion
3.1 RHE Viability
3.2 Testosterone Metabolism
4. Tables & Figures
1 - INTRODUCTION
ADVANCED SKIN AND HAIR, INC. has developed the compound Revivogen Scalp Therapy for hair loss. Male pattern hair loss (MPHL) is a potentially reversible condition in which dihydrotestosterone is an important etiologic factor (Olsen et al, 2006). Dihydrotestosterone, the efficient steroid, is produced from testosterone by 5α reductase. Inhibition of 5α reductase activity is known to improve MPHL.
Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, Wilson T, Rittmaster RS, 2006. The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. J Am Acad Dermatol., 55(6):1014-23
BIOalternatives performed this study in order to assess the effects of the test compound on the metabolism of testosterone in reconstructed human epidermis. This model has been shown to be useful for the evaluation of inhibitors of this metabolism (Bernard et al. 2000; Int. J. Cosm. Sci.,22, 397-407).
The steroid 5α-reductase isoenzymes (5αR) transform testosterone (T) into 17β-hydroxy-5α-androstan-3-one (5-dihydrotestosterone, DHT). This reaction is crucial in the action of androgens.

2 - MATERIALS AND METHODS 2.1 Biological model
Reconstructed Human Epidermis (RHE)
- Tissues: 18 RHE (0.50 cm², 10 days), batch n° 01015-31
- Culture: at 37°C and 5 % CO2
- Culture medium: differentiation medium
2.2 Test compound and references
Test compound Stock-solution Dilution Application
Revivogen Scalp Therapy
batch 07-06 (AD070315/2) Liquid supplied by the study promoter and stored at room temperature. - topical at 50 µl/RHE
References Stock-solution Dilution Application
Finasteride
batch 231664 10-2 M in ethanol In water topical at 10-5 M/RHE
Avodart®
batch 053721A 10-3 M in ethanol In water topical at 10-6 and 10-5 M/RHE
2.3 Testosterone
Testosterone: [4-14C] testosterone (Amersham B76, 54 mCi/mmole, 2.35 nmole/epidermis). [4-14C] testosterone stock-solution was dissolved in ethanol and diluted in sterile water (1% ethanol final).
2.4 Treatment
The RHE were topically treated (or not, control) with the test compound or the references. Three RHE were used for each experimental condition.
After 24h of treatment, the RHE were topically re-treated and incubated for 5 hours. After incubation, the test compound and references were removed from the top of the RHE and 100 µl of the labelled testosterone solution were loaded on the stratum corneum of each RHE (127 nCi/epidermis).
After a 24-hour incubation period, the media underneath the RHE were collected for sterols analysis.
2.5 Extractions and analysis
Transepidermal diffusion assessment: the amount of testosterone that passed through the epidermal tissues was measured by liquid scintillation counting (LKB 1211 Rackbeta counter) of a fraction of culture medium.
Metabolism analysis: the steroid molecules from culture media were extracted by 2 volumes of chloroform/methanol (98:2) and dried. The various molecular species (testosterone metabolites) were separated by thin layer chromatography (TLC) on silica plates (RE/Silice, Whatman) in a solvent system containing dichloromethane, ethylacetate and methanol (85:15:3). The plates were autoradiographed and testosterone metabolites were quantified using a phosphorImager and specific software (Packard instrument).
3 - RESULTS AND CONCLUSION

Schematic simplified pathway for testosterone metabolism.
Effects of finasteride (from Bernard F-X et al., Int. J. Cosmetic Science, 22 397-407 (2000))
Tables 1 and 2
Untreated control:
After 24h of culture, the rate of testosterone metabolism was very high.
Dihydrotestosterone (DHT) was clearly identified in the steroid profile. DHT was the major metabolite in the control epidermis. After 24h, about 74% of the deposited testosterone was converted into DHT. Other important metabolites were androstane-diols (e) and 4-androstene-3,17-dione (b).
Effects of finasteride:
Finasteride at 10-5 M strongly inhibited the transformation of testosterone into DHT (67% inhibition compared to the control). Furthermore, as expected, finasteride decreased the amount of androstane-diols (e) and induced a strong accumulation of 4-androstene-3,17-dione (b) (Figure 1).
Effects of dutasteride:
Dutasteride at 10-6 M and 10-5 M strongly inhibited dose dependently the transformation of testosterone into DHT (respectively 80% and 86% of inhibition of the DHT production compared to the control). Furthermore, as expected, dutasteride decreased the amount of androstane-diols (e) and induced a strong accumulation of 4-androstene-3,17-dione (b) (Figure 1).
Effects of Revivogen Scalp Therapy:
Revivogen Scalp Therapy (5 mg/cm²) strongly reduced the production of DHT (90% of
inhibition of the DHT production compared to the control).
4 - TABLES AND FIGURES
Table 1: Diffusion of [14C]-testosterone (and metabolites) through RHE. Trans-epidermal diffusion (24h)
Treatment Conc cpm sd n % control nmol steroid
Total testosterone - 326060 - 1 - 2,35
Unrelated control - 125307 14977 3 100 0,9
Finasteride 10(-5)M 147747 6335 3 118 1,1
Dutasteride 10(-5)M
10(-6)M 150160
159213 10809
20653 3
3 120
127 1,1
1,1
Revivogen Scalp Therapy 5 mg/cm2 30026 1181 3 24 0,2

Figure 1: Thin layer chromatography and autoradiography of [14C]-testosterone and metabolites after transepidermal diffusion (24h).
Table 2: Effects of Revivogen Scalp Therapy and the reference compounds on the production of testosterone metabolites. Instant Imager analysis of TLC in figure 2 (direct radioactivity measurement)

Figure 2: Effects of Revivogen Scalp Therapy and the reference compounds on DHT metabolism.
[H έρευνα είναι attached στο παρών post]
